 It is singly bonded to 2 oxygen atoms and doubly bonded to 1 oxygen atom. Nitrogen is considered to be the central atom in the nitrate ion. The bond length of the N-O bonds can be given as 125 pm. 
The resonance hybrid of the NO 2 – ion suggests that every oxygen atom holds a partial charge of magnitude with -½. Moreover, the resonance hybrid of this polyatomic ion, which is obtained from its various resonance structures, is used to explain the equal bond lengths, same as illustrated below. Whereas the Lewis dot structures of the NO 2 – ion highlight a difference in the 2 N-O bonds' bond order. 
In the case of nitrite ion, the bond lengths present in both nitrogen- oxygen bonds are equal. A formal charge is used to predict which resonance structures are most favoured. Also, electrons' delocalization can be described through the fractional bonds (denoted by dotted lines) and the fractional charges present in a resonance hybrid.Īt times, the resonance structures are not equivalent, and it is essential to determine which one (s) describes the best actual bonding. The different structures of resonance of the carbonate ion (CO 3 2- ) can be illustrated above. 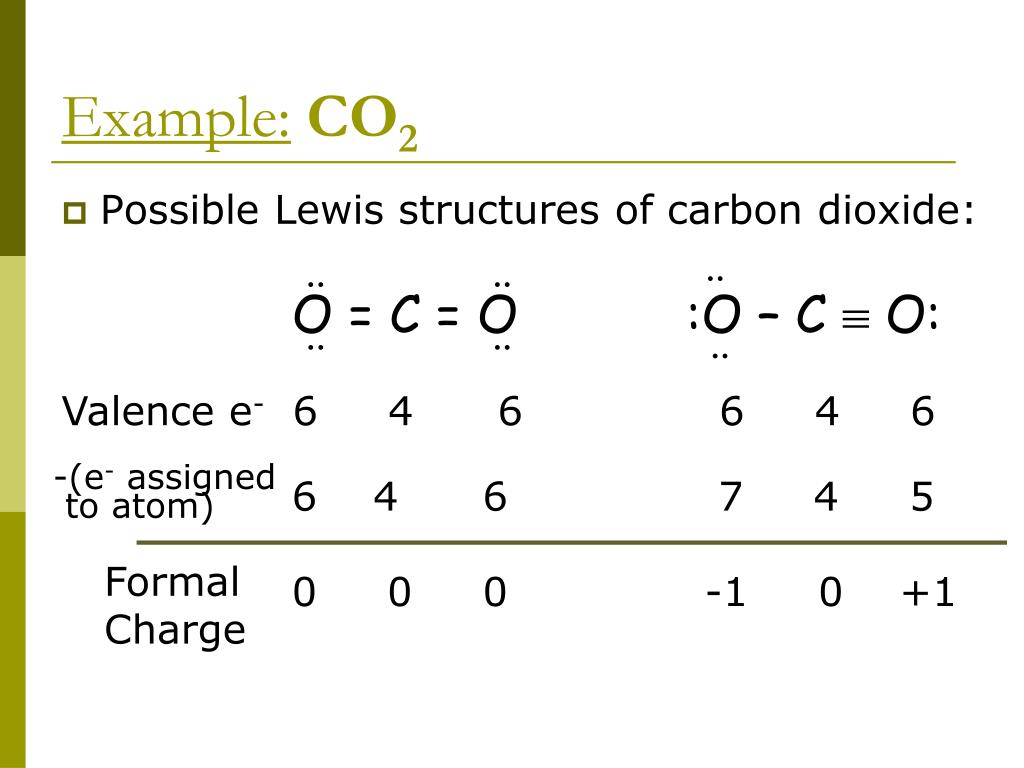
To define resonance in chemistry, the bonding in specific ions or molecules by merging several contributory forms or structures, jointly known as resonance structures or canonical structures within the valence bonding theory, into a hybrid resonance (or the hybrid structure). In such cases, the structures of resonance can be used to describe chemical bonding. In most of the cases, a single Lewis structure has always failed to explain the bonding in either a polyatomic or molecule ion because of the presence of fractional bonds and partial charges in it. Resonance structures are the Lewis structure sets that describe the electron's delocalization in a molecule or a polyatomic ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
